The widespread use of modern drugs has already made it possible to significantly increase the average life expectancy worldwide, and it is predicted that the average life expectancy will only increase in the future. Such achievements would not have been possible without the development of the modern pharmaceutical industry and the continuous improvement of drug therapy methods.
However, for a very long time, the pharmaceutical industry did not have a reliable understanding of the molecular processes in living systems and mechanisms of disease development. In addition, there was no knowledge about specific molecular “targets” that could have an impact on disease control.
The development of the first drugs was not only a trial-and-error method – which is inherent in science as a whole – but this path also did not completely take into account the principles of the structure of living matter at the molecular level.
The scientists and doctors who developed the first drugs worked with some molecules that could affect other molecules, and they had absolutely no idea and could not imagine the details and complexity of these interactions.
Namely, any disease at the molecular level is a consequence of a dysfunction of proteins and/or genes encoding them. The human genome contains about 20,000 genes that encode proteins. The effects of modern drugs are directed to more than 500 targets. At the same time, many diseases are caused by the dysfunction of not one, but at least 5 to 10 related proteins and genes encoding them.
Multiple disorders characterize neurogenerative diseases
Neurodegenerative diseases (NDs) belong to such types of disorders associated with multiple protein dysfunction. The most common NDs are Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and amyotrophic lateral sclerosis. Currently, the world has seen a rapid increase in patients with NDs. According to a report by the World Health Organization, the total number of people with dementia worldwide amounted to around 50 million in 2015, and there are around 10 million new cases every year.
There are no radical treatments for neurodegenerative diseases at present. Current treatment methods only help to improve symptoms, relieve pain, and increase mobility. Therefore, there is a great need for therapies to prevent and slow the progression of neurodegenerative disorders.
The major challenge in drug development against NDs is the fact that we poorly understand the mechanisms of the development of NDs. As mentioned above, NDs are characterized by multiple disorders. Thus, current drug development paradigms have shifted from a “single target” approach to drug development targeting multiple aspects (targets) of the disease. Also, the shift is from treating NDs in the later stages of disease progression to focusing on prevention and neuroprotective strategies at the early stages of disease development.
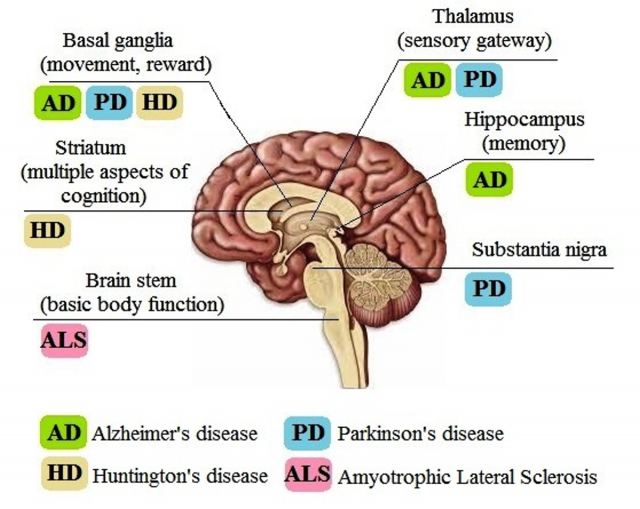
Image credit: Larisa Ivanova
The main obstacles to the development of new drugs against NDs are the long-term asymptomatic development of these disorders, i.e. the clinical symptoms characteristic of each disease tend to appear after almost complete degradation of the key regulatory neurons and the depletion of compensatory brain reserves, as well as the complexity of the brain, its working mechanisms, insufficient availability of resources, and ethical limitations.
Another obstacle is the fact that finding and developing new drugs is a rather time-consuming and expensive process. The total time and money spent on the development of a new drug can range from 12 to 15 years and exceed more than $2 billion.
How computer technology helps drug development
However, in recent decades, the progress in computer technology has had a significant impact on the development of novel drugs. Rational drug design is an essential tool in this area. Rational drug design is the development of new drugs based on knowledge of a biological target. In most cases, the biological target for drug design is a protein that is related to the development of the studied disease.
So, how do recent computer technologies help develop new drugs? A wide variety of methods for the rational drug design allow scientists to select the most preferred and appropriate method for developing a new drug. For example, a sufficient amount of experimental data makes it possible to calculate the biological activity of a compound based on its structure and physicochemical properties. Extensive experimental data also help predict new compounds with the same or improved biological activity. The name of this method is quantitative structure–activity relationship (QSAR).
The next-generation method of rational drug design is based on knowledge of the 3D structure of proteins associated with a studied disease. This approach is called the structure-based drug design. In my work, this approach was used to develop new compounds that mimic the effects of the native substance that forms a complex with the target protein to serve a biological purpose.
How does this method work? How is it possible to create a new active compound, knowing only the structure of the protein? Namely, every single protein in our body, even if it does not work correctly or, on the contrary, does its job too hard, has a so-called active centre through which it binds to other proteins or facilitates a chemical reaction.
The first step of the corresponding design is a thorough analysis of this active centre using molecular modelling programs. It is possible to study in detail which amino acids form the active centre of a protein and how they are located, or to analyse which changes occur in a protein when it binds to other proteins or biologically active substances. Therefore, the selection of small chemical compounds is crucial in order to find out if they bind sufficiently to the active centre of the protein.
Designing a drug is like solving a 10,000-piece puzzle
Imagine a 10,000-piece puzzle: this detail seems to fit well, but something is confusing… The next detail does not fit either… The same thing happens in the design of new drugs. The procedure for selecting the most suitable compound is called molecular docking, or virtual screening in the rational drug design.
Molecular docking programs are concerned with the evaluation of “puzzle pieces” suitable for the studied protein. The molecular docking programs are used to assess how a particular compound binds to the studied protein using the binding energy, that is, how much effort is required to bind (“fix”) the compound to the active centre of the protein (Remember the puzzle! You cannot match the wrong piece no matter how hard you try).
In most cases, compounds (“puzzle pieces”) are taken from public libraries of compounds or designed fragment by fragment by scientists themselves. I used this stepwise construction method to develop a new active compound that mimics the action of a protein named neurotrophic growth factor GDNF.
One of the important functions of neurotrophic growth factors in our body is to keep the neurons alive. The degradation of these factors leads to the death of the neurons and causes neurodegenerative diseases. The GDNF is important for the survival of neurons affected during the development of Parkinson’s disease.
The compound discovered by my colleagues and me showed the ability to directly activate and mimic the action of GDNF. Whereas this compound still has moderate biological efficiency, it is the first compound of this type of activity. As such, it opens completely new prospects in the development of new drugs against Parkinson’s disease.
As mentioned above, there are several approaches in computational drug design, each of which has its advantages and limitations. Thus, using a combination of different drug design methods can improve the accuracy of predictions.

Image credit: Larisa Ivanova
Multitarget drugs affect multiple proteins
In my doctoral work, a combination of two different methods allowed identification of several potential drug candidates among natural compounds against Alzheimer’s and Parkinson’s diseases, as well as neuropathic pain. In the same way, a new potential multitarget agent was developed against two proteins related to Alzheimer’s disease.
Considering the fact that in most cases NDs are associated with the dysfunctions of several proteins, the development of such multitarget drugs will be one of the most promising directions in the development of drugs against neurodegeneration. A drug that regulates the functions of multiple targets reduces side effects, the pharmacological overload of other body systems, and also avoids unwanted effects as a result of drug-drug interactions.
In conclusion, the recent computer technologies play an important role in the search and development of new drugs not only against neurodegeneration, but also for other diseases (cancer, viral diseases, etc.). Virtual screening of large libraries of compounds allows multiple potential drug candidates to be quickly identified, significantly reducing time and money spent. Using other methods of drug design, the possible mechanism of action for newly developed or known drugs on a target can be studied in detail. Close collaboration with biologists allows development of a sufficient theoretical basis for the further successful development of new drugs.

__________________________________________
Larisa Ivanova defended her PhD thesis on the Design of Active Compounds against Neurodegenerative Diseases at the University of Tartu in 2020.
__________________________________________

